Prematurity
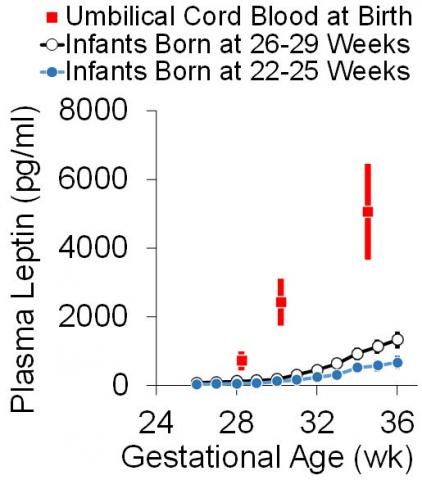
Premature infants are at an increased risk of hypertension, diabetes and obesity. The figure to the right shows the profound leptin deficiency that develops following preterm delivery with postnatal blood levels far below the reference levels obtained from umbilical cord blood at the same gestation. We have now demonstrated improved leptin levels in babies that receive maternal breast milk, and Dr. Roghair has increased efforts to eliminate the inequity in breastfeeding rates that contribute to infant and maternal health disparities.
SSRI Exposure
A significant percentage of pregnant women receive antidepressants during pregnancy. Although short-term side effects are manageable, there is emerging evidence intrauterine SSRI exposure may program adult cardiovascular and behavioral outcomes. Utilizing complementary mouse models and epidemiological studies, we are defining the molecular underpinnings of this “post-SSRI syndrome”. These NIH-funded basic science and clinical investigations are led by Dr. Sarah Haskell.
Cystic Fibrosis
Our proximity to the cystic fibrosis research groups at the University of Iowa led to our observation that the most common cystic fibrosis-causing mutation (CFTR-deltaF508) significantly impacts vascular tone. We are now aggressively assessing the effect of CFTR on arterial function in a number of species, including mice, pigs and humans that carry a copy of the CFTR-deltaF508 mutation.